Neuropreservation of Alcor Patient A-1068
This report is in three parts. The first part is a lay report, the second part is a detailed technical report, and the third part is an essay on some of the bioethical issues involved with this case (“Reflections on a Suspension“).
From Cryonics, April 1985
ALCOR MEMBER PLACED INTO SUSPENSION
by Mike Darwin
Late in January, we received a phone call from the husband of an ALCOR member in Florida informing us that his wife was experiencing some serious health problems and that both of them were planning on returning to their “home” state of Wisconsin for more definitive diagnosis and treatment. Neither the member nor her husband want any publicity, so we’ll assign them the pseudonyms of Paul and Mary Clark here, in order to protect their identity and facilitate relating the events that follow.
Mary Clark had a long history of lymphoma which, until a few months previously, had been in remission. With the reactivation of her cancer, Mary’s health had taken a steep decline and it appeared likely that in the relatively near future she was going lose her 12-year battle with the disease. Paul and Mary have both been cryonicists of long standing and have been intimately familiar with all aspects of the cryonics program. In fact, this was not the Clarks’ first brush with the possibility of Mary’s death and they had taken the precaution of having personnel standing by several times in the past.
This time, however, Mary’s condition seemed far worse. She was already in advanced liver failure by the time she left Florida, and a diagnostic work-up, which included a liver biopsy, indicated that her liver had been almost completely replaced by tumor. Her condition began to deteriorate rapidly and Paul placed a call to us. We had already been preparing for the possibility of flying out to a “remote standby” so, when the pager went off and we learned that one of our members was perhaps only a few hours away from deanimation, we went into high gear. The pager sounded at a little after 10:00 AM on the morning of Friday, February 8th, and by 2:50 PM the same afternoon, Jerry Leaf and Mike Darwin were air borne, en route to the University of Wisconsin Hospital, located in Madison, Wisconsin.
In the cargo hold of the aircraft were six heavy trunks containing resuscitation equipment, stabilizing medications, and a portable blood pump, buble oxygenator and enough premixed perfusate to do a blood washout. Shortly after Mary had arrived at the University of Wisconsin Hospital, we contacted a mortician, Richard Bartell whom we had dealt with on another suspension who was located in a small town about 80 miles southwest of Madison. Mr. Bartell proved to be his usual honest and cooperative self, and suggested that we use the facilities of another firm which he was acquainted with, located just a few minutes from the hospital. Mr. Bartell offered to contact the owners of the firm and act as a reference for us.
As it turned out, the mortuary we were referred to, Fitch, Lawrence and Sanfillippo (which I’ll abbreviate here as FLS) proved invaluable. FLS was the oldest mortuary in Madison, and had moved into brand-new facilities only 11 years previously. The mortuary was very well situated with respect to the hospital, and was, without a doubt, the most intelligently designed facility of its kind that I have ever seen. The preparation room (where bodies are embalmed and cosmetized) was considerably larger than our 16′ x 20′ operating room at Cryovita, and was a well lit, thoughtfully laid out, and sparkling-clean expanse of tile and wood-grain Formica countertopping and cabinetry. In fact, the preparation room at FLS was more spacious and better appointed than many clinical operating rooms I’ve seen! The prep room adjoined a large garage area which could be accessed by automatic overhead doors. In short, the facilities were superb, orders of magnitude better than the cramped, dirty prep rooms I’ve been accustomed to finding in most mortuaries.
The only thing better than FLS’s facilities was its staff. Mr. Sanfillippo is a community leader and fisherman par excellence; his walls are adorned with hundreds of fishing trophies and photos of Mr. Sanfillippo with VIPs from astronauts to Governors competed with the trophies for space. Mr. Sanfillippo proved to a genuinely warm and pleasant man, and his son-in-law Dan Ketelsen, who handles the day-to-day business of running the mortuary, proved to be not only equally pleasant but 110% reliable and cooperative.
We first spoke with Dan by phone about week before our arrival in Madison. We were fairly apprehensive about obtaining the hospital’s cooperation and in making sure that supplies such as ice and oxygen would be on hand to facilitate our transport and washout of Mrs. Clark. We followed up our phone call with a letter of detailed instructions, and Mr. Clark went over to the mortuary to meet Dan and look the place over. Immediately, Dan took control of the situation. He and Sam began negotiating with the hospital, using their respective professional and personal contacts to smooth things over. Arrangements were made with the hospital mortician to facilitate quick release of Mary’s “remains” and to allow us to wait on the premises for deanimation to occur. Dan took over the complicated job of running paperwork around to hospital physicians and lawyers and basically had everything arranged when we arrived on the scene.
Dan met us at the airport upon our arrival, and he and his wife assisted us in loading our trunks into their van. Then they drove us directly to the hospital where we met with Paul to discuss his wife’s condition and prognosis. Our arrival at the hospital found Mrs. Clark to be considerably improved, but still in the Intensive Care Unit in critical condition. At that time Mrs. Clark’s blood pressure was being supported with intravenous medication and fluid. We met with the nursing staff caring for Mary and explained who we were and what “this was all about.” We found the staff to be somewhat guarded and suspicious at first, but this quickly changed. As soon as they realized that we were “medical people” and that we were genuinely concerned about Mr. and Mrs. Clark their attitude changed to one of supportive cooperation.
Since both Jerry Leaf and I work in a clinical environment, we were able to speak the language and share the concerns and frustrations of the nursing staff. I know what my reaction would have been if two strangers had shown up in the middle of the night on my floor with six crates of equipment and a long list of strange requests! The first major issue would be where to put them and their equipment. Hospitals are notoriously overcrowded places — they simply don’t have an inch of storage space to spare, and I’m sure that one of the places they considered putting us at first was out the door. I stated immediately that I knew this would be a problem and that I sympathized with the serious inconvenience our presence was likely to cause. Once we had a good rapport established with the staff they went out of their way to accommodate us. Since it was a weekend and things were quiet, they cleared some space in their supply room and let us station our equipment there.
The next hurdle was to talk with the physician who was following Mrs. Clark in the ICU. He proved to be a very approachable fellow who’s skepticism diminished as he discussed Mary’s case with us and answered questions we asked about her condition and listened to our explanation of what we had planned. Everything was proceeding smoothly, more smoothly in fact than we had dreamed possible.
The following day Mary was still doing well, far better than anyone expected, including her doctors. ICU bed space is valuable, and since Mary was clearly terminal, it was decided to move her to another floor. Two other factors had a part in this decision. First, Mary’s physicians were concerned that she be moved to an unmonitored floor, in other words to a bed where she would not be on a cardiac monitor. They had taken our requests into consideration and were concerned that she would continue to show cardiac electrical activity many minutes after true mechanical cardiac arrest had occurred. This is a common phenomenon in slowly dying patients and is known as an “agonal rhythm.” Agonal rhythm can last as long as 30 to 40 minutes following true cardiac arrest. In Wisconsin, death can be declared either on the basis of cardiac and respiratory arrest or on the basis of brain death. If the patient is on a cardiac monitor, then cardiac arrest cannot be declared until all electrical activity has ceased. If, on the other hand, the patient is not on a monitor, then death can be pronounced on the basis of auscultation or listening for a heart beat and respiration, and checking for absence of a pulse.
Clearly, it would be better for Mary if she didn’t have to be exposed to 30 or more minutes of warm ischemia while we waited for the heart to exhaust all of its metabolic reserves and stop generating electrical signals. The other side of this coin was that someone had to be with Mary more or less continuously to call in the physician as soon as death occurred or appeared imminent. Unfortunately, cryonics personnel cannot serve in this capacity in a hospital setting because of “conflict of interest.” Technically, we stand to “gain” if the patient dies, so we can never be alone with the patient unless a relative or staff member is present. On such short notice, supportive private nursing personnel would be almost impossible to get. So, the task of sitting with Mary fell to Paul.
Another issue to be settled was the kind of supportive care Mary was receiving. Such support had been appropriate early-on in order to give us time to arrive and get deployed. Now, however, it served only to prolong the inevitable. While Mary was not in any pain at this point, being confined to bed with numerous I.V. and arterial lines cannot be described as exactly comfortable. It was decided to discuss with Mary what course of action she wanted to follow since she was fully alert and oriented. Quite sensibly Mary elected to have the medication and I.V. support withdrawn and to “get it over with.”
To everyone’s amazement, this action did not result in immediate cardiovascular collapse. Mary improved somewhat and was able to take food and fluid by mouth. She knew there was a cryonics team standing by, and asked to meet with Jerry and I. My impressions of Mary Clark are difficult to share. I was struck immediately by her great warmth and gentle concern for everyone but herself. Understandably, Mary was very apprehensive about the well-being of her husband of 35 years and we ended our first meeting by her extracting a promise from me to see to it that he had a good lunch and got a little rest. She also joked with me and apologized for “not dying on schedule.” She was genuinely more concerned about the welfare of Jerry and I than she was about her own situation. She seemed reassured by our presence and by the support she knew that we would provide Paul when the inevitable occurred. Her sense of humor and realism about her situation remained intact up to the bitter end. A few hours before deanimating she was remarking on the injustice of finally being able to fly out to California lying down (“first class”) and not having any windows to look out of! Only a cryonicist could keep that kind of balance. When I spoke with her last, by phone, and tried to provide some optimism about her condition, she was quite firm with me that she needed no false hope and that she knew she had very little time left.
After Mary’s move to the Hematology floor, we established a relationship with several new sets of nurses and her attending physician, Mark Bozdick, chief of Hematology/Oncology. Dr. Bozdick took time to sit down and talk to us, and we discussed cryonics, cryobiology and resuscitation medicine at some length. Dr. Bozdick was aware of the long-standing interest of the Clarks in cryonics (this proved to be important, since it defused a suspicion that some of the staff had that we were vultures preying on people’s last minute panic and desperation) and provided a tremendous amount of support and cooperation. Amazingly, we were allowed to “camp out” on sofas in a waiting room a few feet from Mary’s room, and the staff rounded up a Lazy-Boy chair for Mr. Clark to “sleep” on in Mary’s room. So, the vigil began. For three days and four nights Jerry and I were at the hospital almost round the clock. For the first couple of days we took sponge baths in the restroom adjacent to the waiting area, and finally rented a motel room nearby so we could bathe. I even grabbed a few hours of sleep in a real bed at the motel early on, when Mary looked stable, but Jerry stuck with sleeping at the hospital.
Late in the evening of the 11th, Mary began to complain of shortness of breath. Over the next four hours or so her condition deteriorated and at 2:48 AM on the morning of February 12th the physician on-call pronounced Mary Clark legally dead. Within four minutes or so of respiratory arrest, we coupled Mary to an HLR and began administration of transport medications and external cooling.
Intubation and HLR support went smoothly. We were very concerned over the adequacy of gas exchange, because the proximate cause of death was pulmonary edema. Undoubtedly gas exchange was not anywhere where we would have liked it to be, although we were pleasantly surprised when we drew an arterial blood sample on arrival at the mortuary and found that it was bright red. Venous blood was appropriately dark and desaturated of oxygen.
By the time we began the femoral cut-down to couple Mary to the blood pump and oxygenator, her temperature had dropped to 26.5°C. It was here that we encountered the only serious problem of the entire operation. Mary was massively edematous at the time of deanimation. Surgical wounds filled up with fluid as fast as we could sponge it away. The wound behaved like a hole in sand at the beach. No sooner was the field sponged dry than it was full again. Neither Jerry nor I had ever encountered working conditions like this. It slowed down location of the femoral artery considerably. We couldn’t find the right femoral vein. Not only was tissue fluid a problem, but bleeding was a problem. We administered heparin (of necessity) as part of our transport protocol, and the bleeding, which normally would have been manageable was yet another complication overlayed on the deluge of tissue fluid. The bleeding not only contributed extra fluid to fill the wound, but also rendered the tissue fluid opaque. Despite careful extensive searching and expansion of the wound (including assistance from the mortician) we could not find the femoral vein. After about forty-five minutes of further searching, we decided to explore the left groin. We immediately found the femoral vein and slipped a cannula into it. We experienced real difficulty on this side and were forced to shut down the HLR to control the bleeding. Fortunately, by this time Mary’s temperature was under 25°C and we could afford a few minutes of circulatory arrest to facilitate venous catheter placement. We later learned that Mary had had a history of phlebitis in her right leg. Apparently this had resulted in complete obliteration of her femoral vein. This points up the importance of making medical records available to us before death. Had we known of a history of vessel disease in the right leg we would either not have attempted cannulation on that side, or would have abandoned the search for the femoral vein in a more timely fashion. The presence of the edema and the difficult working conditions, coupled with lack of critical information held us back.
Once we had the cannula in place, we were able to go on bypass with a portable blood pump and a bubble oxygenator. We selected a bubbler over a membrane because of simplicity of set-up, the larger reservoir capacity of the bubbler, built-in heat exchanger, and better capability of dealing with clots. We were particularly concerned about clots, because we had been unsure to what extent the hospital would cooperate with us before we arrived on the scene. It was quite possible that there might have been as much as a one or two hour delay in releasing Mary if the hospital refused to cooperate and made us go through “normal channels.” This would have provided more than enough time for clotting to get started. Bubbler oxygenators have a large foam column which we’ve found acts ideally as a pre-filter for larger clots and prevents them from blocking the circuit. As it turned out, clotting was not a problem we had to deal with.
Once we had Mary on the pump, she cooled quite rapidly to 10°C. We then carried out a total body washout with 13 liters of MHP-1, the perfusate we’ve been using in our recent series of dog experiments. We terminated perfusion at a little below 10°C and transferred Mary to a Ziegler case where she was packed in ice from head to toe for shipment to California. Dan Kettelson had thoughtfully constructed a special wooden shipping case to act as a thermal and mechanical protector for the Ziegler. The former was especially important since the temperature outside was considerably below the freezing point. We are especially sensitive to this risk since a patient in the past was inadvertently frozen by morgue personnel where death occurred prior to shipping to California for cryoprotective perfusion. During the six-hour wait for our departure time to roll around, we put the shipping case in one of FLS’s two walk-in coolers to conserve the ice inside. We checked the cooler temperature on a frequent basis with two thermometers, to insure that the temperature was above freezing.
I watched the shipping case containing Mary be loaded onto our plane, and explained to the handling personnel that she should not be dropped or manhandled in the way “corpses” often are. When we arrived at Ontario airport, Al Lopp whisked Jerry Leaf off to Cryovita to get a jump on final preparations for the perfusion, and Hugh Hixon met Paul and I with the van. I went out onto the ramp to assist the lone cargo handler unloading the shipping crate, since it weighed over 450 pounds with all the crushed ice. Within about forty minutes of landing, we were en route to Cryovita.

Scott Greene looks on as the patient’s temperature is checked following her arrival from Madison, Wisconsin, via air freight.

Perfusionist Bill Jameson laying out circuit diagram.

Scrub Nurse Brenda Peters assists lead surgeon Jerry Leaf in gowning for surgery.

Brenda Peters stabilizes the recirculating perfusate reservoir as Jerry Leaf connects it to the heart-lung machine.
I arrived to find Cryovita awash in personnel in surgical garb, and Mary was quickly unloaded from the van and transferred to the staging area behind the operating room. Hugh Hixon had been coordinating activities while we were away and he had done a beautiful job of assembling people and preparing the lab for perfusion. Anna Tyeb, Scott Greene, and Al Lopp had come in during the morning to do a complete clean-up of Cryovita. The operating room, which has been used for the washout experiments on dogs, got a double dose, plus a last-minute scrub-down by Anna, and calibration of the test equipment. We had our usual 100% turn-out of staff. I took a few minutes to assemble everyone and tell them of Mary’s last few minutes. I explained that her deanimation had been difficult and that Paul and the nurses and I were with her at the last holding her hands and comforting her. I told the team that I had told Mary it was O.K. for her to let go now, that we were there to catch her, and that we would not give up, that we could continue to care for her. I asked everyone to give their very best, because she trusted us, and because she was and is a very special person.
Paul had flown out with us from Madison, and once he saw that things were underway and flowing smoothly at the lab, he checked into a motel a mile or so away for some much needed, long overdue rest.
The California end of the operation was an anticlimax. I’m accustomed to perfusions being affairs of incredible tension and tremendous disorganization. Mary’s perfusion was the complete opposite of this. Things went smoothly. The effects of the numerous dog experiments were in evidence everywhere. Note taking was high quality and uniform, personnel didn’t fumble with syringes needles or other “unfamiliar” equipment and everyone felt reasonably self confident. After days of high tension in Madison, Cryovita was almost a let down. Not that I would have it any other way! Oh, for a lifetime of similar “letdowns.” Nevertheless, I was surprised at how calmly and smoothly everything went. For the first time, even though there hadn’t been a suspension in four years, a cryonic suspension was routine, or at least as routine as they ever get.
Mary and Paul had both decided on neuropreservation as the most sensible course to follow. They could have afforded whole-body, but neither felt it a sensible thing. As Paul commented, “I’d thought about preserving just the brain years ago, but didn’t dare open my mouth because I felt there would be such protests against it.” Mary and Paul were quite open about going with the neuro option to the staff at the hospital, and we were all quite surprised to find that while there were many questions related to going “head only” there was little hostility and more than a few comments that it made more sense than taking along a broken-down body. One nurse commented to me that it helped her to better understand what we were after. She said that it forced her to realize that we weren’t just counting on a cure for cancer, but that we expected complete control over life and that we obviously intended to settle for nothing less than a brand-new, healthy body.
This was the first time that we were able to apply a new technique of perfusion which we had heretofore evaluated in animals and found to be superior to perfusion of the head via the carotids. Jerry went in though the chest as would be done for a whole body perfusion, but then tied off all the vessels except the carotids and the vertebrals. This allowed us four points of perfusion for the brain, instead of the two available with just the carotids, and it also allowed us to stop the troublesome problem of “run-off” of perfusate down the vertebrals into the body. This shunting of perfusate away from the brain down a path of less resistance (the open vertebral arteries connect with the carotids via the Circle of Willis at the base of the brain) has been a serious barrier to good perfusion of the brain in the past. The use of the median sternotomy with aortic root perfusion eliminated this problem. We were able to achieve strikingly good circulatory isolation of the head using this approach. At the conclusion of glycerol perfusion, the line of demarcation between perfused and nonperfused tissue was incredibly sharp and began a few millimeters above the clavicle. The contrast was so sharp it was as if someone had drawn a line between the perfused and nonperfused zones with a felt-tipped pen.

Circulatory isolation of the patient’s head was achieved by tying off vessels in the chest and perfusing via the aortic root. This approach allows the brain to be perfused through all four arteries which supply it.

The patient, cooled to dry ice temperature and immersed in silicone cooling liquid, awaits transfer to the TA-60 for further slow cooling to liquid nitrogen temperature.
Glycerolization proeeded very smoothly, with a more or less linear ramp to a terminal concentration which we believe was in the zone of 2.8 M, or about 26%. Precise tissue glycerol values can only be obtained by direct analysis of the tissue, something we clearly cannot do in the case of the brain, so we can only infer terminal glycerol concentration from evaluation of the effluent samples.
Fred and Linda Chamberlain made the long drive down from Lake Tahoe to help out during the perfusion. They came primarily to act as “go-for’s,” but as it turned out, Fred, working with Bill Jameson and Hugh Hixon played a key role in calibrating the glycerol ramp pump and calculating the rate of glycerol concentrate addition. Calculation of addition rate is a task which must be done once the circuit is set up and its volume measured and the mass of the patient’s body or head has been estimated. Fred greatly expedited this process and once again demonstrated his invaluable skills as a team member.
Linda brought in groceries (which the Chamberlains contributed) and then took over monitoring cool-down of the patient in dry ice after everyone else had drifted off to sleep. The following day, Fred and Linda helped Paul prepare for his return flight to Florida, and accompanied me over to the cemetery where Mary’s “mortal” remains would be cremated.
At 8:10 PM on February 14th, Mary was transferred from the silicone cooling fluid where her temperature had been dropped to -77°C to a standard aluminum neuro container which in turn had been nested inside a polyethylene tank. The space between the container and the tank was filled with dry ice. This whole assembly was then transferred into an MVE TA-60 wide-mouthed cryogenic dewar. The TA-60 was closed and lowered via a chain hoist into a dual-patient (whole-body) cryogenic dewar which had a foot or so of liquid nitrogen in the bottom. Over the next 12 days, Mary was very slowly cooled to liquid nitrogen temperature by gradually lowering the TA-60 into the dual-patient dewar and periodically adding liquid nitrogen. At 2:45 AM on the morning of February 26th, Mary was transferred to long term liquid nitrogen storage in our MVE A-2542 multipatient dewar.
Mary will remain with us, fiercely protected and lovingly cared for until the world catches up with her and our needs. Right now she is as safe as our technology and current resources can make her. It’s up to us to carry on and see to it that her trust in us and in the future we’ll help to create, was not misplaced.
From Cryonics, February & March, 1986
NEUROPRESERVATION OF ALCOR PATIENT A-1068
by Michael G. Darwin, Jerry D. Leaf, and Hugh Hixon
INTRODUCTION
On February 8th, 1985 ALCOR was notified that a Suspension Member with a long history of chronic illness was in critical condition at the University of Wisconsin Medical Center in Madison, Wisconsin. According to the patient’s husband, physicians treating the patient expected deanimation to occur within a matter of hours or, at most, days.
A two-man rescue/stabilization team was dispatched to Madison to stand by in the event deanimation occurred. The team was equipped to undertake initial stabilization of the patient with a heart-lung resuscitator (HLR) and carry out total body washout (TBW) prior to transport.
On February 12th, 1985 the patient deanimated and was promptly coupled to an HLR and transported to a local mortuary where TBW was carried out. The patient was then shipped via airfreight to Cryovita Laboratories in Fullerton, California, for cryoprotective perfusion and cooling to liquid nitrogen temperature for long term care.
This report documents the care given this patient and seeks to critically evaluate the results of clinical and laboratory evaluations conducted during and after this suspension. From this evaluation, we hope to improve our techniques for cryonic care.
In keeping with established ethical practices and ALCOR’s and Cryovita’s own standards for confidential treatment of patient’s records and identities, this patient will be referred to here only by her ALCOR number (A-1068).
PAST MEDICAL HISTORY
Past medical history and the admitting examination presented below are summarized from the patient’s medical records provided by the hospital with permission of the patient and her next of kin.
The patient was a 68-year-old caucasian female with diffuse histocytic lymphoma whose history immediately prior to deanimation was one of ascites, abdominal pain, and elevated liver enzymes and bilirubin.
The patient had stage IV diffuse histocytic lymphoma since 1975. She was initially treated with 5,000 rads of radiation to her spine for an epidural mass (which was surgically debulked) followed by systemic chemotherapy with Cyclophosphamide-Oncovin(vincristine)-Prednisone-Adriamycin(doxorubicin) (COPA) until 1977, at which time there was no evidence of disease. In March, 1984, she developed a malignant chylothorax and a mediastinal mass which biopsy disclosed to be diffuse histocytic lymphoma. She subsequently received chemotherapy with COPA and 3,500 rads to the mediastinum. This was followed by sclerosis of her left pleura with tetracycline in an attempt to control pleural fluid accumulation secondary to an obstructed thoracic duct. Her last chemotherapy was in October, 1984, with no evidence of disease at that time.
Three weeks prior to her final admission she developed weakness, abdominal distension, anorexia with weight loss, and light-colored stools. A work-up in Florida, where the patient was spending the winter, revealed elevated BUN, creatinine, alkaline phosphatase, SGOT, and bilirubin. Abdominal computerized tomography (CT) showed hepatomegaly with ascites, but without masses or adenopathy. Paracentesis revealed exudative fluid with suspicious cytology. Because of the likelihood that her disease had recurred, the patient and her husband elected to return to the University of Wisconsin Medical Center to pursue further medical care.
ADMITTING EXAMINATION
Past medical history was remarkable for hypothyroidism and a right lower extremity deep vein thrombosis. The admitting examination, from her medical records, describes a cachectic, chronically ill-appearing white female who was afebrile. Admitting blood pressure was 100/70. Examination of the head, eyes, ears, nose, and throat was remarkable for mild scleral icterus and palsy of the superior oblique muscle of the left eye. A CT scan of the head which was performed to rule out CNS neoplasm was unremarkable. The patient had no adenopathy on examination. Lungs were clear bilaterally. Cardiac examination revealed no jugular venous distension, S1 and S2 with a I/IV systolic murmur. The abdomen was distended, soft, and mildly and diffusely tender without rebound. The liver was 14 cm by percussion, with a firm, smooth, slightly tender edge. The patient had 2+ lower extremity edema. Neurologic examination was without focal findings except the left 6th cranial nerve findings noted above (palsy of the left superior oblique eye muscle).
HOSPITAL COURSE
Due to dehydration, as evidenced by her elevated BUN and creatinine, as well as by her hypercalcemia and hyperkalemia, the patient was vigorously supported with IV fluids and treated with Kayexalate(tm) for her hyperkalemia. This treatment resulted in improved renal function.
Due to the presence of ascites and deteriorating liver function, the patient underwent a laparoscopically-guided liver biopsy and paracentesis. This revealed grossly diffuse, massive replacement of the liver by lymphoma. Following the biopsy, the patient’s course was complicated by a hypotensive episode and she required resuscitation with large amounts of fluid and temporary support of blood pressure with dopamine. The patient subsequently stabilized.
The patient’s prognosis and situation were discussed with the patient and her husband and it was decided that no cardiopulmonary resuscitation would be given prior to pronouncing legal death and that further care would consist of supportive and comfort measures only.
During the approximately 90 hours following her liver biopsy, the patient continued to require IV fluid support to maintain adequate blood pressure at a rate of approximately 400cc per hour. Massive peripheral and systemic edema developed secondary to declining renal and liver function. Despite her continued downhill course, the patient remained alert and fully oriented until the evening of 2/11/85 when she began to experience dyspnea, followed by confusion, loss of consciousness, fall in blood pressure and subsequent respiratory and cardiac arrest. The immediate cause of deanimation was hypoxia secondary to massive pulmonary edema.
RESUSCITATION AND INITIAL STABILIZATION
The patient was pronounced legally dead on February 12, 1985 at 0248 hours CST (deanimation). Within approximately 3 minutes of the last agonal respiration, manual cardiac compression and bag-mask ventilation was begun. The patient was intubated and coupled to a Brunswick HLR 50-90. Following placement on HLR support, intravenous administration of transport medications was begun via Hickman catheter and the patient was packed in Zip-Loc plastic bags filled with finely crushed ice. The patient was afebrile at the time of deanimation with a measured rectal temperature of 36.5øC. Transport medications consisted of: 1.5g sodium pentobarbital, 18g tromethamine, 100g mannitol, 7.5mg verapamil, 2.0mg naloxone, 4.4mg metubine iodide, 210mg cimetidine HCl, 375mg methylprednisolone, and 500ml Dextran40 (10% in 5% dextrose solution). At the time of deanimation the patient was massively edematous with an estimated fluid load of 10 to 15 kg. Transport medications were administered on the basis of the patient’s dry weight. All medications, excepting tromethamine and Dextran40, were given as boluses while awaiting transport from the hospital. Sodium Pentobarbital was administered to secure anesthesia since the patient promptly exhibited return of agonal gasping and spontaneous movement in response to HLR support. Approximately 9g of Tromethamine was given as a bolus and the other 9g was administered over a 40-minute period. Dextran40 was given by continuous IV drip over a 40-minute period.
INITIAL TRANSPORT
After initial stabilization on the HLR and administration of medications the patient was transported to a nearby mortuary for extracorporeal cooling and total body washout (TBW). Cardiopulmonary support and external cooling were continued en route. Upon arrival at the mortuary, the patient was transferred from the transport gurney to an embalming table and repacked in ice. HLR support was not interrupted as a result of the transfer operation. Immediately after positioning the patient on the table a blood sample was collected from an arterial line which was present in the right femoral artery. This and all subsequent samples were collected in 10cc red stopper Vacutainers and refrigerated for later evaluation. Both discard and sample blood were noted to be bright red, indicating arterial oxygenation. Whole blood pH was measured immediately after sample collection with a Horizon model 5995 portable pH meter and was 7.25. Rectal temperature at 0432 hrs CST (deanimation + 1: 44 hrs) was 27.5°C.
FEMORAL CANNULATION
The patient’s right groin was prepared for surgery by swabbing with Betadine solution and draping with sterile towels and a fenestrated drape. The anatomical position of the right femoral artery and vein were located by reference to the pubic tubercle and the anterior superior iliac spine. An incision with a #10 scalpel blade was made at the midpoint between these two structures, beginning at the inguinal ligament and running parallel to the longitudinal axis of the leg for approximately 5 cm.
The femoral artery was promptly identified and an 18 fr. arterial cannula, USCI type 1860, introduced through an arteriotomy and secured with silk ties. Despite extensive dissection which consumed nearly an hour, the right femoral vein could not be located. It was later discovered from the patient’s medical records (which were unavailable at the time of transport) that the patient had a history of thrombophlebitis with a venogram done in December, 1975 demonstrating extreme deep vein thrombosis of the right leg, including the entire right femoral vein.
Owing to lack of success identifying the right femoral vein, the left groin was prepared for surgery and the left femoral vein was promptly raised and cannulated with a 32 fr. venous cannula, USCI type 1967. After location of the left femoral vein, cardiopulmonary support was discontinued for approximately 10 minutes to control fluid accumulation and bleeding during cannulation. The patient’s rectal temperature was 22°C at the time HLR support was interrupted. Both surgical procedures were complicated and greatly slowed by massive fluid accumulation in the wounds due to edema. These complications were particularly difficult to cope with in a field situation where no cautery, suction, or adequate surgical lighting was available.
At 0610 hrs CST (deanimation + 3:22 hrs) cannulation was complete and the patient was connected to a portable extracorporeal circuit consisting of a Sarns modular blood pump, a Shiley S-100A bubble-type oxygenator and a Shiley SF-20, 20 micron filter (See Figure 1 and Plates 1 and 2). Perfusion pressure was measured at the Shiley SF-20 filter, anterior to the arterial cannula, employing an aneroid manometer with a sterile Tri-Med ISOLATER flexible membrane barrier to protect the aneroid from fluid contamination. A calibration curve of measured back pressure vs. measured flow had been generated in advance to account for the pressure increase resulting from cannula-induced flow restriction. This calibration curve was prepared using several cannulae in the size range appropriate for human femoral extracorporeal support.

Figure 1. Portable Extracorporeal Circuit

Plate 1. Portable Extracorporeal Circuit AV=Arterial and Venous lines in sterile package; F=Arterial filter (Shiley SF-20); H=Heat exchanger; M=Manometer; O=Oxygenator(Shiley S-100A); P=Sarns modular blood pump; T=Telethermometer

Plate 2. Portable Extracorporeal Circuit (Closeup). A=Arterial line; F=Arterial filter (Shiley SF-20); H= Oxygenator holder clamp; O= Oxygenator(Shiley S-100A); PL=Pressure line to manometer; S=Stopcock to manometer pressure line; S1= Stopcock to arterial filter bleed line.
The extracorporeal circuit was primed with 1,000 ml Dextran 40 in dextrose, 1,400 ml Plasmalyte, 50 ml 7.5% sodium bicarbonate, and 10,000 units of sodium heparin. Dextran 40 was selected to provide oncotic support in the prime due to its well-documented effects in inhibiting cold agglutination and improving microcirculation in hypothermia (Long, Sanchez, Varco, and Lillehei, (1961); Drake, Macalalad, and Lewis, (1961)). Extracorporeal blood cooling continued at a blood flow rate of 3 liters/minute with a 6 liter/minute oxygen flow rate. Arterial pressure during blood recirculation was 92.5 mmHg. Blood cooling was terminated when the patient’s rectal temperature was 12°C after approximately 20 minutes of perfusion. A blood sample from the arterial line was collected at 0610 hrs CST (deanimation + 3:22 hrs).
Following blood cooling, the patient was allowed to exsanguinate into the oxygenator and the blood collected in this fashion was discarded. The second pump shoe in the circuit was threaded filled with approximately 700 ml of mannitol-HEPES base perfusate (Table 1). Perfusate was sterilized “on line” by filtering into the oxygenator through a Pall 0.2 micron filter. Because the patient had massive interstitial edema and no ultrafiltration or hemodialysis equipment was available it was decided to raise the osmolality of the TBW perfusate from 320 mOsm to 450 mOsm by reducing the volume of water for injection added as diluent to the 4 liters of base perfusate concentrate. Since the TBW perfusion was to be carried out open circuit it was anticipated that the use of hyper-osmolar/hyperoncotic perfusate would reduce the patient’s fluid load by osmotically extracting water from the interstitial and intracellular spaces (Haupt and Rackow, (1982)).
Table 1. Washout Perfusate Composition Component Concentration Mannitol 226.5 mM Glucose 13.3 mM HEPES (Na+ salt) 9.6 mM Calcium chloride 1.3 mM Magnesium chloride 2.7 mM Potassium chloride 37.7 mM Glutathione 6.7 mM Adenine HCl 1.33 mM Hydroxyethyl starch 55 g/l (Colloid Osmotic Pressure=29 mmHg, (Haupt and Rickow, 1982)) Heparin 1000 units/l pH to 7.45 with potassium hydroxide Osmolality: 450 mOsm |
Following completion of the first “pass” of 700 ml, three more passes of base perfusate were perfused (open circuit) with a volume of 2,500 to 2,800 ml each at a temperature of 8°C. A perfusate (venous) effluent sample was collected at 0640 hrs CST (deanimation + 3:52 hrs) near the conclusion of TBW. The patient’s temperature at the conclusion of perfusion was 8°C. The post-TBW hematocrit was unreadable (less than 1%). Examination of the patient following the conclusion of TBW revealed a marked decrease in edema as evidenced by markedly reduced limb girth and reversal of skin turgor which was present prior to TBW. No rigor was evident in the limbs.
PREPARATION FOR AIR TRANSPORT
At the conclusion of perfusion, the right femoral arterial and left femoral venous cannula were left in place and connected with a 3/8″-1/2″ adaptor. The surgical wounds were then closed around the cannula. The wounds were covered in sterile dressing and waterproof plastic adhesive drapes, and the patient was transferred from the embalming table to a metal shipping case containing a rubberized canvas body pouch which had a bed of crushed ice in Zip-Loc bags in the bottom. The patient was then completely covered with ice in Zip-Loc bags in the body pouch and the remaining free space in the shipping case was also filled with ice bags. The shipping case was closed and placed inside a plywood shipping box for air transport to Ontario Airport in Southern California.
ARRIVAL AND PREPARATION OF THE PATIENT
At 2110 hrs PST (deanimation + 20:22 hrs) on February 12 the patient arrived at the perfusion facilities in Fullerton, California. The shipping case was opened and the patient’s pharyngeal and deep tracheal temperature were measured (the latter by placement of a probe via the endotracheal tube). At 2141 hrs PST (deanimation + 20:53 hrs) the pharyngeal temperature was 1.5°C and the tracheal temperature was 3.2°C. An initial examination of the patient conducted at that time disclosed the presence of rigor in the jaws and forearms, its absence in the neck and biceps and the presence of moderate rigor in both lower extremities.
At 2247 hrs PST (deanimation + 21:59 hrs) the patient was positioned on the operating table and the chest, neck, and scalp were prepared for surgery and sterile surgical drapes placed as previously described (Leaf, Federowicz, and Hixon, (1985b)) (Plate 3). At this time a temperature probe was placed in the left choana to more accurately monitor brain temperature. A venous perfusate sample drawn prior to the start of perfusion had a pH of 6.92. At 2336 hrs PST (deanimation + 22:48 hrs) a median sternotomy was begun.

Plate 3. Preparing for medial sternotomy.
SURGICAL PROTOCOL FOR CRYOPROTECTIVE PERFUSION
The basic surgical technique for circulatory access was similar to that employed in previous whole body cryonic suspensions (Leaf, (1981); Leaf and Quaife, (1981)). However, a new surgical approach was employed for vascular isolation of the head for neurosuspension (Fig. 2). In the past, vascular access for cryoprotective perfusion of neuropatients was achieved via bilateral cannulation of the internal carotid arteries. This approach necessitated open circuit perfusion of the head due to contamination of venous effluent with blood and loss of perfusate from the vertebral arteries, which are joined to the carotid circulation via the Circle of Willis. A serious disadvantage of the carotid approach is the possibility of reduced or inadequate cerebral perfusion due to preferential loss of perfusate by retrograde flow through the vertebral arteries. Another potential problem with this approach, particularly in the elderly, is inadequate or absent cerebral perfusion as a result of carotid artery stenosis or thrombosis. A surgical approach which allowed utilization of all four cerebral arteries, i.e., both carotids and both vertebrals, would be more desirable.
In order to achieve such a four point cerebral perfusion the following approach was taken. The sternum was divided medially and a retractor was used to expose the heart. Figure 2 and Plate 4 show the vascular isolation of the cephalic circulation and the position of the arterial and venous cannulae. The arterial cannula was placed in the ascending aorta and arterial perfusate was directed to the brain by ligation of the subclavian arteries with silk ties just distal to the vertebral arteries. An aortic cross-clamp was placed just above the aortic valve to exclude the coronary circulation. The descending aorta was freed from the pulmonary artery by blunt dissection with Metzenbaum scissors. A second aortic cross-clamp was then applied to the descending aorta just distal to the left subclavian artery in order to exclude any arterial circulation to the body. Two purse-string sutures were then placed in the aortic root, one for a 20 fr. arterial perfusion cannula, and the other for an arterial pressure catheter which was connected to a Statham P23Db pressure transducer.

Figure 2. Vascular isolation for neuropreservation.

Plate 4. Cannulae Positions in Median Sternotomy. A=Arterial cannula (20 fr.); C=Aortic cross-clamps; R=Sternal retractor; V=Venous cannula (32 fr.)
The pericardium was opened to expose the right atrium. A purse-string suture was placed in the apex of the right atrium and a 32 fr. venous cannula, USCI type 1967, was advanced through the atriotomy into the superior vena cava. Umbilical tape was passed around the superior vena cava and tied below the cannula tip. In order to prevent contamination of the recirculating system with venous circulation from the extremities, silk ties were placed on the left and right innominate veins just distal to the left and right internal jugular veins. Venous return was collected from a single cannula in the superior vena cava. This approach allowed closed-circuit perfusion of all four cerebral vessels and a gradual addition of glycerol without contamination of the perfusate with systemic blood or perfusate. An additional advantage of this technique was a 50% reduction in cannula cost since only one arterial and one venous cannula were required with this approach.
EXTRACORPOREAL CIRCUIT FOR CRYOPROTECTIVE PERFUSION
The extracorporeal circuit for cryoprotective perfusion is shown in Figure 3. The circuit consisted of two parts: a recirculating system to which the patient was connected and a glycerol addition system (Plate 5). The recirculating system was composed of a sterile 20 liter mixing/recirculating reservoir, arterial (recirculating) pump, an Erika HPF-200 hemodialyzer which was used as a hollow fiber oxygenator (Leaf and Federowicz, (1983)) a Sarns Torpedo heat exchanger, and a Pall EC1440 40 micron blood filter. The mixing reservoir was continuously stirred with a 2″ teflon-coated magnetic stirring bar and a Thermolyne type 7200 magnetic stirrer. Glycerol concentrate was continuously added to the recirculating system with a Travenol 5M1153 hemodialysis pump. Glycerol concentrate was sterilized in line with a Pall PP3802 0.20 micron pre-bypass filter as it was delivered to the recirculating system. The circuit was cut and assembled at the time of use using presterilized lengths of tubing and other components.

Figure 3. Extracorporeal Circuit for Cryoprotective Perfusion.

Plate 5. Pump Rate Check on Glycerol Addition System. CP=Cryoprotectant pump; CR=Glycerol concentrate reservoir
Cultures of the recirculating 5% glycerol perfusate which were taken before the patient was connected to the circuit revealed the presence of a few colonies of the gram negative microorganism Acinetobacter Calcoaecitus Biotype LWOFF1 (commonly know as Mima Polymorpha). This organism is ubiquitous and is found in the soil, on human skin, and on environmental surfaces. The presence of this organism in the perfusate prior to perfusion is indicative of a break in sterile technique either during assembly of the perfusion circuit or collection of the perfusate sample for culture. In the future, preassembled circuits will be used to minimize the chance of bacterial contamination of this kind.
PERFUSATE PREPARATION
Cryoprotective perfusion was begun at 0216 hrs PST (deanimation + 25:28 hrs) on 2/13/85 using 14 liters of 5% w/v glycerol in a mannitol-HEPES base perfusate. Later laboratory evaluation of this perfusate, carried out as part of ALCOR’s quality control operations, revealed that a potentially serious error had been made during perfusate preparation. Personnel preparing the perfusate failed to follow established procedure which calls for the use of a check list to insure addition of all ingredients in the right quantities and appropriate adjustment of pH and osmolality. Failure to use the check list resulted in the omission of calcium chloride and magnesium chloride from the perfusate (these ingredients must be added as a solution which in turn must be prepared immediately prior to use; thus they are not kept with the powdered per pH to 7.72 w/potassium hydroxide fusate components). The composition of Osmolality: 315 mOsm the base perfusate as it was prepared and administered is given in Table 2.
Table 2. Perfusate Composition Component Concentration Mannitol 170 mM Glucose 10 mM HEPES (Na+ salt) 7.2 mM Potassium chloride 28.3 mM Glutathione 5.0 mM Adenine HCl 1.0 mM Hydroxyethyl starch 55 g/l (Colloid Osmotic Pressure=29 mmHg, Haupt and Rickow, 1982)) Heparin 1000 units/l pH to 7.72 w/potassium hydroxide Osmolality: 315 mOsm |
Perfusion with solutions containing less than 50 uM calcium results in loss of calcium from cell membranes, which causes massive edema due to cellular swelling. (Zimmerman, et al, 1967). Fortunately, as later laboratory analysis of effluent samples and recirculating perfusate disclosed, adequate calcium and magnesium were present in the perfusate as a result of their presence as normal impurities in the hydroxyethyl starch employed as a colloid in the perfusate. HES samples from the same lot employed on this suspension have been tested and shown to contain 0.50 mM Ca++ in 6% solutions. The measured arterial perfusate calcium by ionization averaged 0.46 mM, and venous perfusate showed an average of 0.48mM, which are well above the 50 uM danger level. The perfusate contained 5.5% HES, which correlates well with these arterial and venous ionized calcium values.
PERFUSION CONDITIONS
Arterial perfusate temperature averaged 13.0 + 1°C and a flow rate of 500 cc/min was maintained throughout perfusion. Arterial perfusion pressure was 30-50 mmHg. Patient tracheal, sinus, and frontal brain temperature during the course of perfusion and cephalic isolation are shown in Figure 4.

Figure 4. Temperatures During Perfusion.
Paired arterial and venous samples were drawn at 15 minute intervals during perfusion for determination of pH and glycerol concentration, and for later evaluation of electrolytes and other chemistries. Arterial and venous pH during perfusion are shown in Figure 5. The venous pH was persistently lower than the arterial. Despite oxygenation and perfusate flow rates far above those achieved in the past (Leaf, Federowicz, and Hixon, (1985b)) acidosis remained a problem. As in recent suspensions (Leaf, Federowicz, and Hixon, (1985b)), it was thought that the venous pH remaining persistently below the arterial pH was probably indicative of active anerobic metabolism, and it was expected that oxygenation would correct this problem. However, the problem persisted despite oxygenation.

Figure 5. pH During Perfusion.
The acidosis experienced by this patient may in part be due to inadequate levels of HEPES in the perfusate. Foreman et al have demonstrated good control of pH during hypothermic organ perfusion employing HEPES as the buffer at approximately twice the concentration used on this patient (Foreman, Pegg, and Armitage, (1985)). Evaluation of increasing the buffer concentration, use of another buffer with a higher pK, or active electronic control of pH is needed in an animal model in an attempt to solve this problem.
Corneal flattening was present as a normal post-TBW/postdeanimation change prior to the start of perfusion and became more pronounced, proceeding to frank ocular flaccidity after the first 15 minutes of perfusion. During the last 20 minutes of perfusion ocular volume rebounded to preperfusion levels and immediately prior to the termination of perfusion the eyes were firm and the corneas were undimpled, probably reflecting intraocular fluid accumulation and/or edema.
CRYOPROTECTANT ADDITION
Four liters of the 5% w/v perfusate were flushed open circuit through the patient’s cephalic circulation and discarded, after which the circuit was closed and recirculation was begun at a flow rate of 500 cc/min. After 28 minutes of recirculation, 6 liters of perfusate were drained from the circuit, discarded, and replaced with 6 liters of fresh 5% w/v glycerol-containing perfusate. At 0259 hrs PST (deanimation + 26:11 hrs), 7 liters were drained from the perfusate reservoir and replaced with fresh 5% glycerol perfusate.
Linear, gradual addition of glycerol to the recirculating system was begun at 0300 hrs PST (deanimation + 26:12 hrs) with a precalibrated pump, using a 50% w/v glycerol solution in mannitol-HEPES base perfusate (also prepared without calcium chloride or magnesium chloride). Electrolyte and other solute concentrations were kept constant on a molar basis at all glycerol concentrations. The rate of addition of glycerol to the recirculating system was calculated prior to initiation of cryoprotective perfusion by estimating the fluid load represented by the patient’s head and adding that to the known volume of the extracorporeal circuit. A relatively large volume recirculating reservoir (as contrasted with past suspensions) was used to minimize the effects of errors in estimating the patient’s fluid contribution, i.e., to act as an osmotic buffer during glycerolization.
Previous animal studies carried out by ALCOR and Cryovita have demonstrated a serious problem with very stable stratification or “layering” of cryoprotective concentrate as it is introduced into the recirculating system. To avoid this problem, the recirculating or “mixing” reservoir was continuously stirred using a bar-type magnetic stirrer as described above.
The progresS of glycerolization was monitored by osmometry (Precision Systems MicroOsmette Model 5004 freezing point osmometer) to provide, where necessary, corrective negative feedback to the calculated ramp.
A terminal venous perfusate glycerol concentration of 3.0 M was sought with an additional objective that arterial/venous (A/V) glycerol concentration difference be held to less than 100 mM throughout perfusion. Glycerol concentration over the course of cryo-protective perfusion is shown in Figure 6. The objective of maintaining the A/V glycerol difference to 100 mM or less was achieved throughout most of the perfusion. Unfortunately, due to the development of edema perfusion had to be terminated before the target 3.0 M glycerol concentration was reached in the venous perfusate.

Figure 6. Glycerol Concentration During Perfusion.
Cryoprotective perfusion lasted for 158 minutes. Perfusion was terminated at 0454 hrs PST (deanimation + 28:06 hrs) as a result of cerebral edema. Venous glycerol concentration (extrapolated) at the conclusion of perfusion was approximately 2.8 M.
TREPHINATION
After cryoprotective perfusion was begun, an incision was made in the scalp over the left parietal lobe. A burr hole was made as previously described (Leaf, Federowicz, and Hixon, (1985b)) (Plates 6 and 7) to observe changes in cerebral cortical volume and evaluate the degree of blood washout achieved during prior TBW perfusion. The cortical surface was visualized at approximately 0315 hrs (deanimation + 26:27 hrs). Since the burr hole was not made until after the start of cryoprotective perfusion it was not possible to evaluate the degree of cerebral volume reduction (dehydration) the patient experienced during the start of glycerolization. At the time the cerebral cortex was first visualized it appeared normal in volume and free of any blood-filled vessels (Plate 8).

Plate 6. Hudson cranial drill with 9mm burr used to open hole in cranium.

Plate 7. Trephanation with Hudson cranial drill. C=Cranial drill; D=Self-adhesive sterile drape; R=Weitlaner self-retaining retractor.

Plate 8. Burr hole in skull. B=Burr hole; C=Cerebral cortical surface; R=Weitlaner retractor.
Near the conclusion of per-Perfusion the burr hole began to leak clear fluid. Perfusion pressure remained constant and it was felt that this leakage represented weeping of fluid from the cortical surface, the meninges, the dura, or all three, rather than rupture of a cerebral vessel. This phenomenon has been observed primarily in ischemic ally injured cats, and to a lesser degree in nonischemic cats being glycerolized to 3.0 M (Federowicz and Leaf, (1983)). In the cat, the development of such burr hole drainage is close ly correlated with rapid progression of cerebral edema and is usually not observed until after a venous glycerol concentration of 2.0 M or greater has been reached.
Within 20 minutes after significant drainage from the burr hole was first noted, cerebral edema had developed to the point that the cortical surface was beginning to bulge against the margins of the burr hole. If perfusion was temporarily discontinued, the cortical surface subsided to several millimeters below the margin of the burr hole. Conceivably, despite the development of cerebral edema, perfusion might have been continued in an “on-off” mode. However, due to the increasing rate of leakage of fluid from the burr hole it was decided to terminate perfusion. The flow rate of fluid from the burr hole near the end of perfusion was in excess of 5 cc/min.
After the conclusion of perfusion the stainless steel disc of a YSI Type 729 implantable thermistor probe was placed on the surface of the brain, and the burr hole was filled with bone wax. The wound was then closed using interrupted 2-0 Tycron suture and the probe anchored as previously described (Leaf, Federowicz, and Hixon, (1985b)) (Plate 9).

Plate 9. Burr hole closure. P=Probe lead wire; S=Suture line.
GROSS EFFECTS OF GLYCEROLIZATION
Despite the development of interstitial edema, cellular dehydration of the skin and other structures of the head and neck was very evident, even at the conclusion of perfusion. Within 3 to 5 minutes of the start of perfusion the skin of the patient’s face and neck developed the translucent, amber mottling typical of human glycerol perfusions (Federowicz, (1973)). Marked facial dehydration developed as the glycerol concentration was further increased. Efforts to minimize or eliminate these effects by carrying out glycerolization at 13°C to 14°C as opposed to 6°C to 8°C as has been done in the past (Leaf, Federowicz, and Hixon, (1985b)) were unsuccessful.
One interesting feature of this dehydration was the development of a very sharp line of demarcation between the perfused and unperfused areas of the patient’s skin. At the conclusion of perfusion the zone between amber, perfused skin, and normal appearing unperfused skin was as sharp as if it had been scribed with a pen. This is evidence of the effectiveness of the vascular isolation techniques employed on this patient in confining perfusion to the cephalic circulation.
SURGICAL PROTOCOL FOR CEPHALIC ISOLATION
Following termination of cryoprotective perfusion a circumferential skin incision was made at the base of the neck extending anteriorly and posteriorly to just below the margins of the clavicle. The skin was dissected free from underlying connective tissue up to the level of the 5th cervical vertebra to form skin flaps. The muscles of the neck and other structures were then severed with a #10 scalpel blade down to the junction of the 5th and 6th cervical vertebrae. A Gigli saw was passed under the vertebral column and a cut was made between the 5th and 6th cervical vertebrae, which freed the head from the body.
Skin flaps were then closed over the stump of the neck using a skin stapler, after the edges of the flaps were first approximated using interrupted 2-0 Tycron sutures. Because the perfusion cannulae were in the thoracic vessels no decannulation was required before cervical transection. This resulted in a savings of several minutes of surgical time. Use of the Gigli saw also proved superior to the previously employed technique of separating the cervical spine with a Satterlee amputation saw (Leaf and Quaife, (1981)).
TISSUE SAMPLE COLLECTION
At 0736 hrs PST (deanimation + 31:48 hrs) on 2/13/85 the sternotomy wound was extended, a laparotomy performed, and dual samples of the following tissues were collected using clean, but nonsterile technique: spinal cord, heart, liver, intestine, skin, lung, spleen, kidney, and muscle. The tissue samples were placed in 5 ml Nunc Cryotubes where they were glycerolized at room temperature in 2.0 M glycerol in mannitol-HEPES base perfusate. Similar samples were taken and fixed in 4% formaldehyde in normal saline neutralized with calcium chloride for subsequent histological evaluation. The tubes containing the samples were placed in a weighted plastic bag (to prevent floating) and were transferred to the cooling bath containing the patient at 0803 hrs PST (deanimation + 32:15 hrs) (bath temperature of -12°C).
COOLING TO DRY ICE TEMPERATURE
At 0621 hrs PST (deanimation + 29:33 hrs) the patient (head) was enclosed in two polyethylene bags and submerged in an insulated bath containing 20 liters of Dow-Corning 5 centistoke silicone fluid (Silcool) (Figure 7). The Silcool had been precooled to a temperature of -6°C. The frontal sinus, oral, and cerebral cortex temperatures at the start of cooling were 13.0°C, 13.0°C and 11°C respectively.
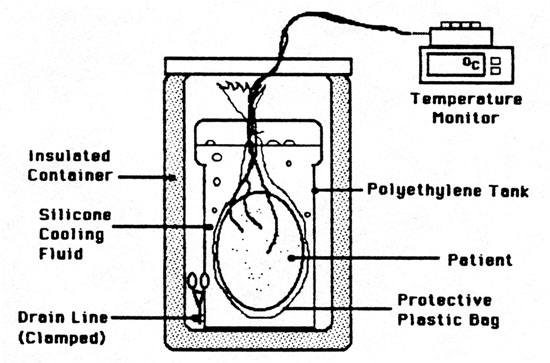
Figure 7. Dry Ice Cooling Bath.
A surface-to-core temperature differential of no more than 10°C -+2°C was maintained at all times once the patient’s core temperature went below 0°C. Due to the patient’s undesirably high temperature at the conclusion of cephalic isolation a differential of nearly 20°C was considered acceptable at the start of cooling in order to rapidly reduce the core temperature of the head.
Figure 8 shows the patient’s frontal sinus, frontal cortex, and oral temperature during dry ice cooling. Cooling to -77°C required 28.5 hours. Silcool, which was employed clinically for the first time during this suspension, performed as expected with excellent retention of clarity (allowing observation of the patient and samples for ice formation) and low viscosity over the entire temperature range of dry ice cooling. No leakage of the fluid into the plastic bags containing the patient was noted.

Figure 8. Temperature during dry ice cooling.
COOLING TO LIQUID NITROGEN TEMPERATURE
Because of serious problems with tissue fracturing which have occurred in the past (Federowicz, Hixon, and Leaf, (1984)), it was decided to cool this patient as slowly and with as small a differential between surface and core temperatures as possible. While it is unlikely that such a maneuver would eliminate the occurrence of fractures, it was hoped that it might reduce their frequency and severity.
After dry ice cooling the patient was removed from the Silcool bath and transferred to a -50°C chest-type freezer, the floor of which was covered with a bed of dry ice. In this environment the protective polyethylene bags were removed and (using gloved hands) the patient was rapidly transferred to a heavy “flannel texture” polyester pillow case. The pillow case containing the patient was then lifted by the “tail” of excess material and the patient was positioned inside a heavy-walled aluminum “neurocan” containing Dacron wool as packing to protect against mechanical shock. The neurocan had previously been nested inside a polyethylene tank with the space between the two filled with dry ice. The lid of the neurocan was closed and covered over with additional dry ice.
The polyethylene tank containing the patient was then placed inside an MVE TA-60 cryogenic dewar. The TA-60 was closed and placed on a support platform of 3/4″ plywood which could be lowered or raised on a chain hoist. The TA-60 was then lowered into a modified MVE A-9000 dual patient storage dewar to which 160 liters of liquid nitrogen had previously been added (Figure 9).

Figure 9. Apparatus for cooling to -195°C.
Over the following 12 days the assembly containing the patient was slowly lowered toward the liquid nitrogen in the bottom of the A-9000. An effort was made to hold the surface-to-core temperature differential to no more than 10°C throughout cooling to -196°C. Cooling to -196°C was carried out over a period of 294.5 hours (Figure 10).

Figure 10. Temperature during LN2 cooling.
TRANSFER TO LIQUID NITROGEN STORAGE
When the patient’s core temperature reached -176°C the TA-60 (still containing the patient) was removed from the A-9000 and filled with liquid nitrogen to a level 6″ from the bottom. The patient’s core temperature was then allowed to drift down to -192°C at which time the TA-60 was slowly filled with liquid nitrogen.
When the patient’s core temperature reached -196°C the neurocan, which was full of liquid nitrogen, was transferred to a styrofoam work box where final preparation of the patient for long term storage was carried out. Final preparation consisted of adding additional Dacron wool packing and proper identification of the patient by attachment of identifying stainless steel tags to the cloth bag containing the patient and to the lid of the neurocan. Thermocouple lead wires were stowed inside the neurocan and the lid was secured in place with stainless steel wire.
The patient was then transferred to ALCOR’s MVE A-2542 storage dewar where she was immersed in liquid nitrogen for long term cryogenic care.
LABORATORY EVALUATIONS
In an effort to evaluate the patient’s physiological condition during resuscitation, extracorporeal support, total body washout, and cryoprotective perfusion, samples of blood and arterial and venous perfusate were collected as often as feasible. These samples were later subjected to analysis for glycerol concentration and electrolyte, tissue enzymes, and metabolite levels.
Clinical chemistries were performed on a SMAC-2 autoanalyzer by a reputable veterinary clinical laboratory. Following analysis, two sets each of 3 ml aliquots of the samples were pipetted into 5 ml Nunc tubes, cooled to -40°C in a mechanical freezer, further cooled to liquid nitrogen temperature and then transferred to the neurocan containing the patient.
CHEMISTRIES: RESUSCITATION AND TOTAL BODY WASHOUT
Results of chemistries on blood and perfusate samples collected during resuscitation, extracorporeal cooling, and TBW are shown in Table 3. Sodium, potassium, chloride, and total calcium levels are shown in Table 4, and are consistent with the patient’s history and present no anomalies.
Table 3. Blood and Perfusate Chemistries
Sample Normal Resusci- Pump End of End Washout
Chemistry Range tation Cooling Washout Resuscitation
(r)
________________________________________________________________________
Albumin 3.3-4.5 g/dl 1.1 g/dl 0.5 g/dl 0.1 g/dl .091
Alkaline 87-250 IU/L 690 IU/L 361 IU/L 10 IU/L .014
Phosphatase
Amylase* 100 IU/L 100 IU/L 100 IU/L (1.0)
Bilirubin <0.5 mg/dl 3.2 mg/dl 1.6 mg/dl 0.1 mg/dl .031
Blood Urea 8-20 mg/dl 50 mg/dl 43 mg/dl 19 mg/dl .380
Nitrogen
Cholesterol 120-330 mg/dl 105 mg/dl 48 mg/dl 0 (0)
Creatine 15-57 IU/L 107 IU/L 93 IU/L 5 IU/L .047
Phosphokinase
Creatinine 0.6-0.9 mg/dl 1.8 mg/dl 1.8 mg/dl 0.5 mg/dl .278
Lipase* ---- 2.2 Neph. 1.5 Neph. 0.8 Neph. (.364)
Units Units Units
Total 6.6-7.9 g/dl 2.4 g/dl 1.7 g/dl 1.2 g/dl ---
Protein**
SGOT 8-20 IU/L 432 IU/L 10 IU/L 52 IU/L .120
SGPT 9-24 IU/L 118 IU/L 119 IU/L 7 IU/L .059
_____________________________________________________________________
Abbreviations: g-grams; dl-deciliter; mg-milligrams;
mEq-milliEquivalents; IU/L-International Units per Liter;
SGOT-Serum glutamic-oxaloacetic transaminase;
SGPT-Serum glutamic-pyruvic transaminase.
Sampling times after deanimation: Resuscitation-1:37 hrs;
Pump cooling-3:22 hrs; End of TBW-3: 52 hrs.
Normal ranges from Hamilton, H. K., Ed., Diagnostics, Springhouse
Publishing Co., Springhouse, PA, 1984, pp1076-83.
*Amylase and Lipase levels shown here reflect the low end
sensitivity of the SMAC-2 and are not accurate.
**Total protein by SMAC-2 is inaccurate in the presence of HES.
|
Table 4 Blood and Perfusate Electrolytes
Sample Normal Resusci- Pump End of Perfusate
Range tation Cooling Washout Concentr.
Chemistry
_____________________________________________________________________
Calcium 4.4-5.0 mEq/l 3.2 mEq/l 2.6 mEq/l 0.8 mEq/l 0.6 mEq/l
Chloride 100-108 mEq/l 113 mEq/l 107 mEq/l 85 mEq/l 47.0 mEq/l
Potassium 3.8-5.5 mEq 10 mEq 8.2 mEq 24.9 mEq 37.7 mEq/l
Sodium 135-145 mEq 129 mEq 126 mEq 76 mEq 9.6 mEq/l
_____________________________________________________________________
Abbreviations: g-grams; dl-deciliter; mg-milligrams;
mEq-milliEquivalents;
Sampling times after deanimation: Resuscitation-1:37 hrs;
Pump cooling-3:22 hrs; End of TBW-3: 52 hrs.
Normal ranges from Hamilton, H. K., Ed., Diagnostics, Springhouse
Publishing Co., Springhouse, PA, 1984, pp1076-83.
|
Electrolytes
As a result of fluid support during her agonal course and the administration of over 1,500 cc of non-electrolyte-containing fluid during resuscitation and transport (i.e., 500 cc each of mannitol, tromethamine, and Dextran 40) sodium, calcium and chloride levels are predictably below normal. The markedly elevated resuscitation potassium (K+) of 10 mEq/L is typical of hypoxia/ischemia. The presence of these elevated levels of K+ also reflects some inadequacy of HLR transport in supporting metabolism well enough to allow for reabsorption of lost intracellular K+. Despite 3 hours and 22 minutes of HLR support in the presence of moderate hypothermia, serum K+ levels were still nearly two times the accepted clinical maximum. Ischemia was already present before the patient deanimated, due to the massive edema evident in the extremities and low perfusion pressure (30 – 50 mmHg 50 to 60 minutes prior to deanimation). An additional factor in elevated serum K+ is the administration of hypertonic and hyperoncotic I.V. solutions, i.e., mannitol, tromethamine, and Dextran 40 which are known to elevate serum K+ (Makoff et al, (1970)).
The reduction of electrolyte levels during extracorporeal support probably reflects the effects of further hemodilution from the 2400 cc prime present in the extracorporeal circuit. In particular, slight reduction of the K+ concentration from 10 mEq/L near the end of HLR support to 8.2 mEq/L is almost certainly due to hemodilution since the rapid induction of deep hypothermia as a consequence of extracorporeal heat exchange would probably have precluded any metabolic reabsorption of K+ (Messmer, Brandel, Reulen, and Nordmann, (1966)).
At the conclusion of TBW, sodium, calcium, and chloride levels had, as expected, declined to values intermediate between prewashout serum levels and levels present in the perfusate. The relatively low levels of extracellular K+ at the termination of TBW compared to the K+ concentration present in the perfusate suggests a need to raise perfusate K+ levels closer to those present intracellularly. Larger volumes of flush solution may also be necessary to impose the desired electrolyte milieu on the extracellular space, particularly in cases where massive systemic edema is present.
Tissue Enzymes and Metabolites
The patient’s extremely low resuscitation levels of serum albumin (1.1 g/dl vs. 3.3-4.5 g/dl normal value) and total protein (2.4 g/dl vs 6.6-7.9 g/dl normal value) reflect both hemodilution and her severe liver disease. The moderate elevation of resuscitation BUN (50 mg/dl) and creatinine (1.8 mg/dl), in the presence of such marked hemodilution is indicative of emerging renal failure which again is to be expected considering the patient’s hepatic status and overall disease burden.
Resuscitation levels of SGOT (432 IU/L), SGPT (118 IU/L), and bilirubin (3.2 mg/dl), even in the presence of significant hemodilution, do not adequately reflect the massive compromise in hepatic function present in this patient. As subsequent autopsy and histological study disclosed, there was little remaining hepatic parenchyma to be injured and thus release enzymes. Pathological and histological examination of the patient’s liver revealed over 90% replacement of the hepatic parenchyma with lymphomatous tissue. Reasons for the patient’s abnormally low cholesterol levels, in probable order of importance are: hemodilution, hepatic insufficiency, and poor nutritional status prior to deanimation. Elevation of alkaline phosphatase and creatinine phosphokinase levels are probably a direct consequence of the malignancy and the hypoxia/ischemia the patient experienced prior to, during, and after deanimation. Unfortunately, laboratory data on the level of these enzymes prior to deanimation is not available to us. Serious predeanimation hypoxia and reduced perfusion were evidenced by the presence of nail bed and limb cyanosis in the patient beginning nearly 24 hours prior to deanimation.
The effect of TBW on the levels of these metabolites and enzymes was profound and indicates, as does the unreadable hematocrit, the efficiency of TBW in removing formed elements and high molecular weight compounds from the vascular space. By contrast, levels of BUN and creatinine were only reduced by approximately 2/3rds. Tissue enzymes, which have a larger molecular weight and which do not move freely across the cell membrane (except as a consequence of cell lysis or injury) were reduced to very low levels, or to levels below the low-end sensitivity of the SMAC-2. The reduction or virtual elimination of the concentrations of these enzymes to very low levels is positive evidence of the integrity of the overwhelming majority of the patient’s cells at the conclusion of TBW. Failure to similarly reduce the levels of BUN and creatinine is consistent with movement of these comparatively low molecular weight compounds from the relatively large reservoir represented by the intracellular and interstitial fluids into the vascular fluid (perfusate).
The values for amylase, lipase, and total protein are inaccurate and reflect a limit on the low end sensitivity of the SMAC-1 and the interfering effects of HES on the total protein test. If the end of washout value for a given compound divided by the resuscitation value of the same compound is denoted as r (see Table 3) then a comparison of the high molecular weight vs. the low molecular weight compounds shows a mean value of 0.329 -+.072 versus a mean value of 0.0603 -+.039, a difference which is significant at the p<0.001 level.
ELECTROLYTES: CRYOPROTECTIVE PERFUSION
As a result of the high concentrations of glycerol present in the perfusate samples collected during glycerolization, it was not possible to subject them to analysis for tissue enzyme or metabolite levels. A preliminary study to evaluate the accuracy of the SMAC-2 in determining electrolyte concentrations in the presence of high concentrations of glycerol was made using both the SMAC-2 and flame photometry. Values obtained in this fashion were found to agree well with each other and the remaining analyses were conducted using the SMAC-2.
Of special concern was the concentration of ionized calcium in the recirculating perfusate during glycerolization. As can be seen in Figure 11 the SMAC-2 determination showed a terminal calcium level of less than 0.5 mg/dl. While this is an adequate level of total calcium to prevent injury due to elution of cell membrane calcium (Zimmerman et al, (1967)), it is below the threshold of reliability of a clinical test. For this reason ionized calcium levels using the ion-specific electrode technique were run on both terminal perfusate and burr hole samples. Calibration runs using glycerol containing calcium solutions were first performed to establish the reliability of this technique in the presence of multimolar concentrations of glycerol. Additional tests were conducted on base perfusate from the same batch which had not been perfused (quality control samples) and finally on a hydroxyethyl starch solution of the same concentration employed in the perfusate. As can be seen from Figure 12, the calcium levels in both the burr hole and the recirculating cryoprotective perfusate were well in excess of the 50 uM/L threshold below which membrane damage is known to occur.

Figure 11. Calcium concentration during perfusion (colorimetric method).

Figure 12. Calcium concentration during perfusion (ion-specific electrode method).
The levels of sodium (Figure 13), chloride (Figure 14) and phosphorus (Figure 15) during the course of perfusion reflect continued decline in the tissue concentrations of these electrolytes. This is due to their dilution in the recirculating system as a consequence of the continual addition of cryoprotective concentrate (which had a lower concentration of these agents than the tissues).

Figure 13. Sodium concentration during perfusion.

Figure 14. Chloride concentration during perfusion.

Figure 15. Phosphate concentration during perfusion.
Conversely, the concentration of potassium (Figure 16) in the recirculating perfusate consistently increased as the depleted intracellular levels of potassium were restored.

Figure 16. Potassium concentration during perfusion.
The final venous reading on all of the cryoprotective perfusion electrolyte evaluations is spurious. The sample from which these evaluations was derived was collected, in error, immediately after the perfusion pump had been turned off. We know from animal work conducted in our own laboratory and in others, using an isolated head model similar to the one employed on this patient, that an accurate venous sample cannot be obtained after the pump is shut down, presumably as a result of seepage of fluid from the rest of the patient’s circulatory system into the venous drainage. We include this datum as a point of information and as a caution to others who may use this technique in the future, since it demonstrates the profound distortion of data which taking samples after perfusion has terminated can result in.
GENERAL DISCUSSION AND CONCLUSIONS
Given the constraints of space, it is impossible to fully discuss the tremendous wealth of logistic, clinical, and physiological information which was obtained from the cryonic suspension of this patient. We will thus confine ourselves to issues which bear directly on the care of future suspension patients.
MEDICAL RECORDS
The authors have previously repeatedly emphasized the need for access to the patient’s medical records as long in advance of carrying out a suspension as is practical. Much of the delay and difficulty encountered with this patient could have been completely avoided had we only had access to this patient’s records. During the three and a half days we waited for deanimation to occur we could have been thoroughly briefed on every aspect of this patient’s prior medical history, and thus have avoided the loss of over an hour of surgical time in futile pursuit of a femoral vein which had long ago been obliterated by disease — in an episode well documented in this patient’s medical records. As a consequence, the patient suffered an unnecessary additional hour of low-quality HLR support as well as greatly increased risk of systemic bacterial contamination due to poor preparation of the alternate surgical site (of necessity we carry only limited quantities of sterile disposable drapes and prep supplies).
It is often difficult to convince the next of kin or the attending physicians of the importance of providing medical records before deanimation occurs. Often, suspension members and patients fail to appreciate the enormous potential for complications and harm which can arise from lack of such information. This case should serve to illustrate the importance of obtaining and utilizing medical information before deanimation occurs.
EXTRACORPOREAL SUPPORT AND TOTAL BODY WASHOUT
Where possible, extracorporeal support should begin in the hospital or nursing home prior to transport. This will require the development of portable, gurney mounted, self contained perfusion units incorporating a blood pump, membrane oxygenator, and heat exchanger. The inadequacy of HLR support, particularly in the presence of pulmonary edema or reduced gas exchange from pulmonary malignancy is likely to be a recurring problem. The logistics of transport, often over considerable distances, to a mortuary or other facility result in too much HLR time with resultant poor cardiac output and slow heat exchange using surface cooling.
When field TBW is undertaken in the future it is highly desirable that two blood pumps be available. Considerable delay and inconvenience resulted from having to thread and rethread recirculating and perfusate filtration pump shoes into the pump head. The presence of an additional pump as well as a hemodialyzer and circuit would also allow for intensive ultrafiltration of the edematous patient during pump-supported cooling. Early correction of edema will almost certainly result in better tissue perfusion during transport and TBW, as well as better cryoprotective equilibration during cryoprotective perfusion. To this end we have upgraded our remote standby kits to include two blood pumps and appropriate hemodialysis equipment for hemofiltration.
A major barrier to prompt initiation of extracorporeal support of this patient was the presence of massive fluid accumulation in the surgical wounds, coupled with poor lighting. Obviously it is impractical to transport operating room lights or other heavy fixtures to the site of remote TBW operations. However, a variety of portable, battery operated head lamps such as those used by spelunkers and outdoorsmen are available. We have purchased two Model 04419 electric headlamps manufactured by Justrite Manufacturing Company of Mattoon, Illinois and have found them very satisfactory in trials.
Controlling fluid accumulation in the wound due to bleeding or drainage of interstitial edema into the wound can only be effectively achieved by electrically powered suction equipment. We rapidly exhausted all available gauze and sterile absorbent material during attempts to raise this patient’s femoral vessels and finally had to resort to use of nonsterile suction equipment (which was kept to the margin of the wound) provided by the mortuary. We have since added suction equipment to our remote standby kit to cope with future episodes like this one.
CRYOPROTECTIVE PERFUSION
Aside from the previously discussed problem with bacterial contamination, the extracorporeal circuit and surgical approach employed during glycerol perfusion were extremely satisfactory. As previously noted, we were surprised by the precise demarcation of glycerolized from unglycerolized tissue. This sharp and uniform demarcation was present not only cutaneously, but also in the other tissues of the neck as well; unglycerolized tissue which was especially soft and gelatinous in consistency due to edema was separated from firmer, dehydrated, glycerolized tissue by a zone only several millimeters in thickness.
Significant cellular dehydration coupled with the development of interstitial edema during glycerolization remains a major barrier to adequate cryoprotection of patients. We attempted to circumvent these problems by providing a slower rate of increase in glycerol concentration and by using a colloid which animal research has demonstrated provides excellent oncotic support, even in deep hypothermia (Leaf, Federowicz, and Hixon, (1985a)). It was also anticipated that edema might be less of a problem in this patient due to the absence of vascular obstruction resulting from cold agglutination and postmortem clotting.
To some extent this last anticipation was justified. In the past, typical perfusion flow rates during whole body cryonic suspension operations have been in the range of 700 to 800 cc/min with perfusion pressures (70-80 mmHg) being the limiting factor. In this patient, a cephalic flow rate of 500 cc/min was maintained consistently with a mean perfusion pressure of 30 mmHg. In other words, perfusion flow to this patient’s head was approximately 2/3rds of the maximum tolerated flow to the entire body of previous suspension patients who were not subjected to prompt extracorporeal support and field TBW.
The failure of maintaining a narrow spread between arterial and venous glycerol concentrations (100 mM or less) to ameliorate cellular dehydration and prevent the development of interstitial edema argues for reevaluation of glycerol as the cryoprotective agent of choice. Since extending the length of the glycerol introduction ramp was not effective in reducing or eliminating these undesirable effects it would seem prudent, at least until a replacement agent can be found, to increase the rate of glycerolization. Increasing the rate of glycerolization would minimize patient exposure time to cryoprotective agent and might conceivably allow a higher terminal glycerol concentration to be reached before edema becomes limiting. An exponential rate of concentration increase should be roughly optimal for reasons indicated by Meryman elsewhere (Meryman and Williams, (1985)).
An urgent priority is the evaluation and selection of a cryoprotective agent or mixture of agents which better penetrates cells and which does not compromise capillary membrane integrity. Previously discussed problems with assessing cryoprotective agent concentration in real-time via in-line refractometery remain unresolved primarily due to the expense of in-line refractometers adequate for this type of application.
TECHNICAL EXCELLENCE
As can be deduced from this technical report, even though we placed into the field two of the most experienced personnel ever dispatched to recover a suspension patient, and provided the most sophisticated technical support package ever deployed, there are a number of areas in which improvement is needed. Where economically feasible, we have acted to make such improvements.
More attention needs to be paid to post perfusion temperature control during cephalic isolation. In the future, whenever possible, perfusion temperature should be lowered toward 0°C at or near the conclusion of the cryoprotective ramp. The burr hole should be closed as quickly as possible, preferably immediately before the conclusion of perfusion so that the head may be adequately refrigerated during cephalic isolation by completely packing it in ice.
In order to reduce the risk of future errors in perfusate preparation a system employing not only a checklist, but second person verification has been instituted. Weighing of perfusate components now must be done by two individuals; one who performs the weighing operation and a second who verifies it. This two-person policy applies also to addition and mixing of perfusate components as well as adjustment of final pH and osmolality and cryoprotectant concentration. The use of such safeguards should virtually eliminate such potentially disastrous errors in perfusate preparation as occurred in this case. As soon as economically practical we will acquire analytical equipment which will allow for quality control checks on perfusate prior to its administration.
In terms of cost-effectiveness we were able to offer a high degree of technical sophistication and yet stay well within budget requirements. This was due largely to the outstanding cooperation and support of Cryovita Laboratories and the support of an all-volunteer suspension team. We wish to extend our sincere thanks to Cryovita Laboratories and to the ALCOR Cryonic Suspension Team whose professionalism and dedication contributed so greatly to the high quality of care this patient received.
REFERENCES
Drake, C. T., F. Macalalad, and F. J. Lewis, (1961), The Effect of Low Molecular Weight Dextran Upon the Blood Flow During Extracorporeal Circulation, J. Thoracic and Cardiovascular Surg. (42, 735 (1961))
Federowicz, M. (1973), Perfusion and Freezing of a 60-Year-Old Woman, Manrise Technical Review, (3, 7-13 (1973))
Federowicz, M., H. Hixon, and J. Leaf, (1984), Postmortem Examination of Three Cryonic Suspension Patients, CRYONICS, (#50, 16-28 (Sept, 1984))
Federowicz, M. and J. D. Leaf, (1983), Cryoprotective Perfusion and Freezing of the Ischemic and Nonischemic Cat, CRYONICS, (#30, 14 (1983))
Foreman, J., D. E. Pegg, and W. J. Armitage, (1985), Solutions for the Preservation of the Heart at 0°C, J. Thoracic Cardiovascular Surg., (89, 867-71 (1985))
Hamilton, H. K., Ed., (1984), Diagnostics, Springhouse Publishing Co., Springhouse, PA, 1984, pp1076-83
Haupt, M. T. and E. C. Rackow, (1982), Colloid Osmotic Pressure and Fluid Resuscitation with Hetastarch\, Albumin, and Saline Solutions, Critical Care Medicine, (10(3), 159-162 (1982))
Leaf, J., (1981), Case Study: KVM Suspension, CRYONICS, (#13, 8 (Aug, 1981))
Leaf, J., and M. Federowicz, (1983), Experimental Hollow Fiber Oxygenators for Organ Preservation Research, Cryobiology, (20(6), 714 (Dec, 1983))
Leaf, J., M. Federowicz, and H. Hixon, (1985a), Asanguineous Perfusion of Dogs at 5°C for 4 Hours, With Long Term Survival, Cryovita Laboratories and ALCOR Life Extension Foundation, Paper presented to the Society for Cryobiology, Madison, WI, June 20, 1985
Leaf, J., M. Federowicz, and H. Hixon, (1985b), Case Report: Two Consecutive Suspensions, A Comparative Study In Experimental Human Suspended Animation, CRYONICS, (6(11), 13 (Nov, 1985))
Leaf, J., and A. Quaife, (1981), Case Study of Neuropreservation, CRYONICS, (#16, 21 (Nov, 1981))
Long, D. M., L. Sanchez, R. L. Varco, and C. W. Lillehei, (1961), The Use of Low Molecular Weight Dextran and Serum Albumin as Plasma Expanders in Extracorporeal Circulation, Surgery, (50, 12 (1961))
Makoff, D. L., J. A. Da Silva, B. J. Rosenbaum, S. E. Levy, and M. H. Maxwell, (1970), Hypertonic Expansion: Acid-Base and Electrolyte Changes, American J. Physiology, (218(4), 1201-7 (Apr, 1970))
Meryman, H. T. and R. J. Williams, (1985), Basic Principles of Freezing Injury to Plant Cells: Natural Tolerances and Approaches to Cryopreservation in Cryopreservation of Plant Cells and Organs, K. K Kartha, Ed., CRC Press, 1985, pp14-47
Messmer, K., W. Brandel, H. J. Reulen, and K. J. Nordmann, (1966), Elektrolytveranderungin in tiefer Hypothermie, III. Beziehungen zur biologischen Uberlebenszeit bei Kunstliehem Kreislauf, Pflugers Archiv (288, 240-261 (1966))
Zimmerman, A., W. Daems, W. Hulsemann, J. Snyder, E. Wisse, and D. Durrer, (1967), Morphological Changes of Heart Muscle Caused By Successive Perfusion With Calcium-Free and Calcium-Containing Solutions (Calcium Paradox), Cardiovascular Research, (10, 201-9 (1967))
REFLECTIONS ON A SUSPENSION
From Cryonics, July 1985
by Mike Darwin
During the recent remote stand by/suspension of an ALCOR member in Madison, Wisconsin we were faced with a new, and at times anxiety provoking situation: the confrontation between cryonics and medical ethics. The issue of where what we’re doing fits into a dying patient’s medical care is one we’ve often talked about in the abstract in these pages, but due to the infrequency of suspensions and hospital cooperation, rarely confronted in reality.
A key issue early on was whether or not it would be legally and ethically possible for the hospital to cooperate with us. Would cooperation imply tacit approval of what we were trying to do? Was cryonic suspension something health care providers had any role in supporting? Initially, things looked good for hospital cooperation. But our optimism quickly turned to anxiety when we learned that the hospital “bioethicist” had been brought in to decide if what we were planning to do involved “human experimentation.” If it did, then it would as a minimum need approval of the hospital’s human experimentation committee and perhaps approval from any local or federal government agencies who might have authority in the matter of human experimentation.
What would the bioethicist and the hospital board decide? Did cryonics constitute human experimentation or merely a postmortem procedure such as an autopsy or embalming? Considering the sad history of bioethics in medicine and the strong inclination of people not “involved” in the problem to make a name for themselves or to “carve out territory” by rendering a negative opinion, there were considerable grounds for pessimism.
As an overlay to the bioethics issue, one of the patient’s nurses, a young, intelligent RN whom I’ll refer to here as Gary, became extremely concerned about the impact of the procedure on the patient’s care and the degree to which the patient was aware of the possible risks of such an “experiment.” Almost from the start, Gary was preoccupied by the ethical issue of a possible “successful” resuscitation. What, Gary wanted to know, would happen if the patient reestablished a normal heart beat and respiration as a result of our application of the heart-lung resuscitator? What if she regained or demonstrated signs of consciousness during resuscitation? His concerns have a basis in fact, since patients who have suffered a cardiac arrest do sometimes regain consciousness during CPR and do sometimes spontaneously convert to a normal heart rhythm during resuscitation activities.
Gary was deeply concerned about the “ethical” question involved here. In his estimation, the patient at that point would be legally and morally alive — in other words, still his responsibility as a health care provider and thus once again outside our area of competence and/or control. How did we plan to deal with these issues? Were we planning on “giving the patient anything to stop such recovery from happening?” The corollary question was, of course, if we were planning on “giving something” (poisoning with potassium was something Gary suggested) would that be murder?
From our perspective as cryonicists it’s easy to find Gary’s concerns quaint or absurd. In a clearly terminally ill patient (who in this case had made the decision herself to terminate all life supporting measures — including even I.V. fluids) it would be pointless to proceed with normal resuscitation (i.e., discontinuing cryonics procedures) only to have the patient suffer more deterioration and “die” again a few hours or days later. But Gary, and presumably the rest of the world, might not see it that way.
As a matter of fact, we have had patients demonstrate some signs of motor activity during “postmortem” HLR transports. We have had patients swallow, shiver, and demonstrate other motor activity normally associated only with “living” people. Because of this we have, for some time, administered massive doses of medications both to protect the patient from any awareness of resuscitation, cooling, and transport, and to provide protection against cerebral anoxia/ischemia by placing the brain into a coma (this is a standard medical maneuver used to treat anoxic and ischemic brain injury). From our standpoint this kind of action makes both good ethical and practical sense. We know that it is possible to restore a patient to consciousness after death is pronounced and that it would be of no benefit to us or to the patient to do so. (It will become even more possible, indeed even probable that our actions could result in restoration of consciousness as our resuscitation and support techniques improve, such as with the application of total cardiovascular and respiratory support with blood pumps and membrane oxygenators.)
From a cryonics standpoint the “ethical issues” are not that we are restoring someone to consciousness or potential consciousness and then proceeding to freeze them. To us, such issues are absurd; after all, the medical profession has the same ability to restore people to life for a few more hours or days in the same situation, but chooses not to do so in order to let people die and disappear forever! Does this make them murderers? Rather, the issue to us is protection of the patient from suffering discomfort or damage while trying to save his/her life. We look at administering medications and proceeding with a suspension in exactly the same way a surgeon would view the application of anesthesia during an operation: as a humane and necessary “extinguishment” of consciousness which works to the benefit of both patient and physician.
Nevertheless, we would be foolish to expect noncryonicists to accept such an argument. We told Gary that there were medications given in the course of the transport protocol that would prevent the patient from waking up and experiencing more suffering. He was not really satisfied with this, and pressed both Jerry Leaf and I for additional information. Gary was also very forthright in questioning us about the limits to which (we felt) he could legally go to in frustrating the suspension, and what in fact we would do if someone did attempt to block suspension of the patient.
One of our greatest fears was that Gary and the bioethicist would get together. It is hard to assess Gary’s motives objectively. At the time (and even now) I feel he was genuinely concerned about the possibility of our procedures inflicting more pain on a patient he could clearly see was suffering and whom he genuinely cared about. I think that Gary also felt a real alienation or displacement of his role as a health care provider. Over and over again he stated that he was troubled by the issue of where his responsibilities left off. What did this cryonics business mean and how did it interface with his duties and responsibilities to the patient?
I learned from talking with other nurses that Gary was a vocal and frequent opponent (often acting as the patient’s advocate) of the kind of useless and degrading medical heroics that some doctors will subject and even psychologically coerce their patients into. In his conversations with both Jerry and me, Gary repeatedly brought up the issue of “at what price life?” Was what we were doing really a decent and human thing to do? Or, was it a depersonalizing and degrading attempt to defeat death at the cost of the patient’s dignity and humanity?
It’s not hard to see how people who are not cryonicists could feel this way. Even under the best of circumstances, the physical procedures involved in perfusing and freezing someone are pretty cold, clinical, and easy to interpret as “degrading.” In this patient’s case there was the added issue of “neuropreservation” which could also be seen in the same light.
As things developed, the bioethicist did not come up with any “hard and fast” opinions. He said that the decision as to whether or not cryonic suspension constituted “human experimentation” and thus merited a full review by the hospital ethics and human experimentation boards hinged to a large extent on whether or not one thought it would work. If the position was taken that what we call a patient is in reality a cadaver, then ethics, at least “ethics in medicine” had little or nothing to do with it.
Since the hospital already had started to cooperate there was substantial medical and ethical pressure for them to continue. Moving the patient to another facil- ity would have been extremely trying both logistically and emotionally, especially in view of the fact that death was a few hours or at most days away. Also there was the question of finding another facility that would accept such a “problem.” No doubt these were issues that coursed through their minds. Added factors in our favor were that the medical staff had established a reasonable working relationship with us (and felt we were not kooks and that we were scientifically and technically knowledgeable), and the patient and her family had flawless legal preparations, firm intent and a long history of interest in, and commitment to, cryonics.
It could easily have gone the other way if any one of those factors were missing or there was dissent from any quarter to cloud the issue or raise red flags. Had Gary gone to the press, demanded action by the ethics committee or otherwise attempted to derail things, the patient might well not have been suspended under the relatively good conditions she was or even suspended at all.
I have no way of knowing for sure why Gary didn’t take the action he was no doubt contemplating, to block the patient’s suspension. Perhaps he realized that this was something that this patient and this family wanted. Or, perhaps, in his conversations with us he realized both that we were genuinely concerned about the patient and that we had iron wills which would not abide of easy interference. There was one very tense moment, where Jerry Leaf and Gary squared off, and Jerry, with cool, razor steel in his voice told Gary he would sincerely regret interference with this suspension if he attempted any. That moment was perhaps a test of wills, and we won.
We won, perhaps because we were and are willing to go a step further. For “them,” the noncryonicists of the world, interfering with us is born (in most instances) from rather “idealistic” concerns. I am not seeking to minimize here the ease with which we or others can be hurt by misplaced idealism or the so-called good intentions of other people trying to mind our business for us. But I would point out that when push comes to shove it is, after all, a matter of life and death for us. Perhaps at least one of those noncryonicists out there got the idea (maybe for the first time ever) that when push comes to shove we’re ready to push and shove — with a vengeance.
